What is a Clinical Trial?
A clinical trial is a study in real people. We compare a new intervention – like a test medicine, procedure, or device – to an old one or a placebo so that we can learn how that intervention affects us. Often, people in studies will be put into certain groups that either get the test intervention or the old one or the placebo so we can see the difference between them.
Some Words to Know
Healthy Volunteer
A Healthy volunteer is a person with no known major health problems who joins a clinical trial to test a new drug, device, or intervention.
Inclusion and Exclusion Criteria
Inclusion and Exclusion Criteria are the Protocol rules about which people can join the study. Each study will have different rules. Inclusion Criteria are those things a person must have to be included (maybe a diagnosis of Diabetes Type 2, or an age of over 18). Exclusion Criteria are things a person cannot have (possibly another diagnosis, like high blood pressure or the taking of a certain medication).
Informed Consent
The Study Team must inform you of the risks and potential benefits of the clinical trial before you can truly decide to join or not. They will give you an Informed Consent Form with these details and answer all your questions. You can then decide to say Yes or No.
A placebo is something that contains no medicine, like salt water. We often use them in studies when there is no existing therapy for a disease. Sometimes, as in cancer studies, we test a new medicine idea to be given at the same time as an existing therapy. So people will be given their regular medicine plus the test medicine, OR their regular medicine plus a placebo. We compare the placebo to our new therapy idea.
We might want to learn if the new treatment leads to more side effects compared to a placebo, or if the new treatment helps people heal faster than the placebo. In COVID vaccine studies, researchers wanted to know if people given a new vaccine had lower, higher, or the same chance for example of getting COVID as those that got a placebo. Comparing them helps us see if our new ideas benefit our patients.
Principal Investigator
In clinical trials, the Principal Investigator is the leader of the study. They are in charge of watching the health of the study Subjects and making sure that the study plan is followed.
Protocol
A Protocol is the carefully designed plan for a study or clinical trial. It describes how the researcher will keep participants safe and find the answers to their scientific questions.
Randomization
Randomization is a way that Subjects get assigned to a comparison group in a clinical trial. It means that whether a Subject gets put into Group A or Group B is random, or like a flip of a coin.
Subjects
People who volunteer to join a research study are called Subjects.
Phases of a Clinical Trial
Each new approved medicine that becomes available in the United States has been studied in people. If a new treatment idea looks promising in the laboratory, researchers still have to prove that it will help humans, know at which dose, and know what any side effects might be so that we can be informed as patients.
New medicine ideas to be tested are called “investigational drugs”. Researchers have to write a detailed plan to test their investigational drug in humans, and submit their plan to the Food and Drug Association (FDA), before they are allowed to begin.
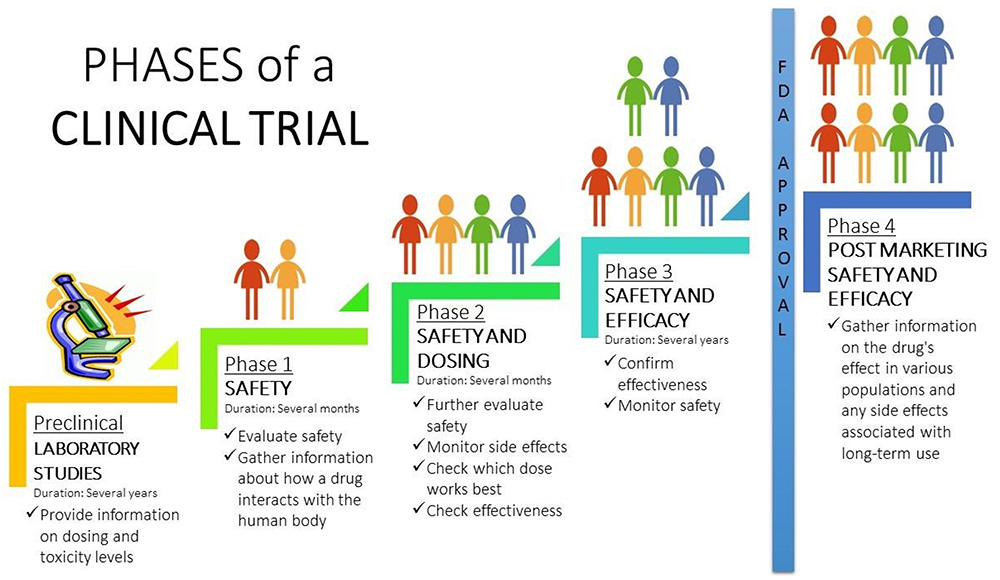 (Click image to view larger)
(Click image to view larger)Phase 2: These studies enroll typically hundreds of patients to see if the drug is safe, and to better understand the right dose for people. Researchers will also look at data to see if it may be effective as a treatment.
Phase 3: These studies enroll hundreds or thousands of patients, and collect lots more information on the effectiveness of the drug, and its safety.
Important: If at any time the investigational drug proves to be ineffective or too harmful, the researcher will not be permitted to continue to the next studying phase.
If after finishing Phase 3 studies, the researcher believes the new drug idea works well in people, they must submit a New Drug Application to the (FDA) with all the information they learned. The FDA will review and decide if the new drug can be made available to the public.
Phase 4:These studies look more deeply at an approved medication, sometimes to compare it to another one to learn which will work better for you, or to get more information about side effects over time.