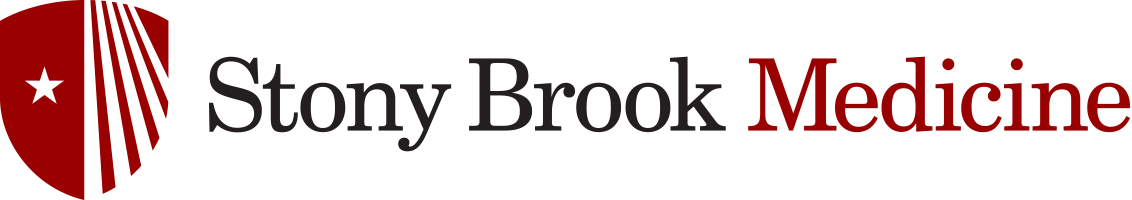SPERM RETRIEVAL FOR ASSISTED REPRODUCTIVE TECHNOLOGIES
Our Male Infertility Specialist:
Sperm quality in obstructive and non-obstructive azoospermia
In the normal male reproductive tract, sperm exiting the testis have minimal motility and limited egg fertilizing capacity. Sperm acquire the potential for improved motility and fertilizing ability during epididymal transit. So, in the unobstructed epididymis, sperm of optimal quality (as evaluated by percent motile cells) are found in the most distal epididymis. The obstructed epididymis shows the opposite pattern of sperm quality: optimal sperm quality in the proximal epididymis and very poor quality in the most distal segments. This finding of "inverted motility" is expected in the obstructed male reproductive tract since sperm production continues in the testis and reabsorption of those sperm is an active process in the most distal regions of the system. The most distal obstructed epididymis tends to contain dilated yellow tubules that are packed with macrophages reabsorbing old, degenerated sperm. Therefore, sperm retrieval should be performed from the proximal obstructed epididymis.
In non-obstructive azoospermia (testicular failure) some sperm production is at least focally present within the testis, despite the fact that inadequate numbers of sperm are released from the testis to contribute to the ejaculate. A common observation for testicular sperm samples is that retrieved spermatozoa are immotile or have a sluggish twitching motion. After several hours of incubation in vitro, testicular sperm typically show some motility. The lack of initial motility does not necessarily reflect a lack of viability for testicular sperm, since these sperm have never acquired motility. Non-motile ejaculated sperm have acquired and lost motility, as sperm viability is lost, rendering the sperm useless for ICSI.
Typical criteria for a poor prognosis of sperm production, including an elevated serum FSH level or decreased testicular volume, does not prospectively predict which patients can have sperm found with testicular sperm extraction (TESE). Even the histologic patterns on diagnostic testicular biopsy (Sertoli cell-only, maturation arrest, hypospermatogenesis) cannot definitively predict the chance of finding sperm with TESE. However, many centers require diagnostic biopsies prior to TESE procedures to rule out carcinoma-in-situ (intratubular germ cell neoplasia) that is present in up to 3% of men with NOA who are candidates for treatment with TESE-ICSI.
The goals of sperm retrieval are a) to obtain the best quality sperm possible, b) to retrieve an adequate number of sperm for both immediate use and cryopreservation [where possible], c) to minimize damage to the reproductive tract so as not to jeopardize future attempts at sperm retrieval or testicular function. For men with NOA, sperm retrieval has most commonly been performed with an open extraction procedure that allows sampling of large volumes of testicular parenchyma, since the concentration of sperm is so low in the testis.
For men with obstructive azoospermia, a variety of techniques for sperm retrieval have been successfully applied.
Percutaneous techniques.
(All pictures: Dr. Peter Schlegel, Cornell Medical Center, with permission)
Percutaneous Epididymal Sperm Aspiration (PESA)
PESA has been advocated as superior to open microsurgical approaches in that it can be performed without surgical scrotal exploration, it is repeated easily at low cost, and it does not require an operating microscope or expertise in microsurgery. The procedure can be performed under local or general anesthesia. The testis is stabilized and the epididymis is held between the surgeonís thumb and forefinger. A 21-gauge butterfly needle attached to a 20-ml syringe is inserted into the caput epididymis and withdrawn gently until fluid can be seen entering the butterfly needle tubing .The procedure is repeated until adequate numbers of sperm are retrieved. PESA yields very small amount of epididymal fluid and contamination with blood cells is frequent. For the 10-20% of attempts where sperm are not retrieved with PESA, open sperm retrieval or percutaneous testicular aspiration is possible.
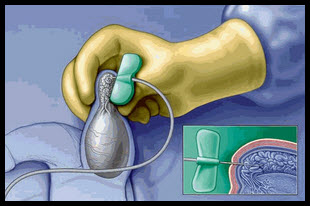
Percutaneous Testicular Sperm Aspiration
The technique of Testicular Fine Needle Aspiration (TFNA) of the testis was initially described as a diagnostic procedure in azoospermic men. Subsequently, testicular fine needle aspiration or biopsy for the recovery of spermatozoa has been described. Percutaneous puncture and aspiration of the testis can be performed using a 21-23 gauge needle connected to a 20 cc syringe in a Menghini syringe holder .
Systematic TFNA was used to localized areas of sperm production in the testis ( testicular mapping) in small number of patients with nonobstructive azoospermia as a guide for subsequent testicular sperm extractions.
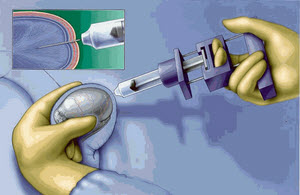
Percutaneous testicular biopsy
Percutaneous testicular biopsy is a technique that combines the effectiveness of testicular sperm extraction (TESE) with the minimally invasive nature of TFNA to improve sperm yield with a procedure that is rapid and simple. A large core (14-gauge) biopsy gun with a short (1 cm) excursion can be used (Microvasive ASAP channel-cut biopsy system, Boston, Mass.) . As with TFNA, testis is stabilized between the thumb and index finger of one hand. The biopsy gun is directed diagonally along the longitudinal axis of the testis, entering on the anterior surface of the lower pole, aimed toward the rete testis. A biopsy is obtained by firing the biopsy gun with the tip firmly applied to the testis through the overlying skin.
The limited published experience to date with TFNA makes critical evaluation of this technique difficult, although it is evident from our experience that 1) sperm retrieval is routinely possible with TFNA for men with obstructive azoospermia, 2) occasional hematoceles and hematomas are possible with this technique.
The advantages of percutaneous aspiration techniques are that they can be performed with less anesthesia, without open scrotal exploration and its attendant postoperative discomfort, and without microsurgical expertise. The percutaneous techniques are easily repeatable as well as potentially less expensive than microsurgical techniques and do not require special microsurgical training. However, lower pregnancy rates have been published after percutaneous sperm retrieval (24%) compared to that achieved with microsurgical epididymal sperm aspiration (36-82%) with ICSI (24% vs. 36-56%). In addition, the inconsistent ability to retrieve adequate numbers and quality of sperm for cryopreservation with percutaneous approaches is a disadvantage relative to open sperm retrieval techniques.
Although fine needle aspiration has been reported as a technique of sperm retrieval for NOA, men have not been adequately characterized with formal testicular biopsy before these procedures, and it is possible that some of the patients thought to have NOA were actually obstructed. Fine needle aspiration would be much more likely to retrieve sperm in men who are obstructed and may make results of treatment unevaluable unless diagnostic biopsies were performed.
Open surgical sperm retrieval
Microsurgical Epididymal Sperm Aspiration (MESA)
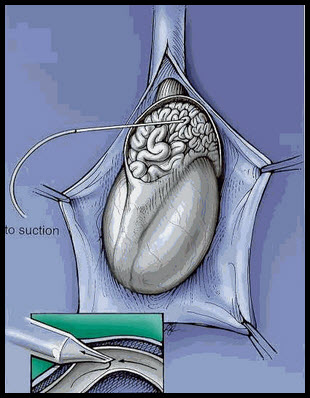
Epididymal sperm retrieval was initially performed using open, microsurgical technique commonly referred to as microsurgical epididymal sperm aspiration (MESA). This technique offers the advantages of dissection of individual epididymal tubules under direct vision, meticulous hemostasis to minimize sperm contamination with blood cells and aspiration of multiple sites to retrieve large numbers of sperm with optimal quality to allow immediate sperm use for assisted reproduction as well as sperm cryopreservation. To avoid contamination of sperm by blood cells, a technique of micropuncture was developed. Individual tubules are cleanly punctured, as occurs during venipuncture for blood drawing. With gentle aspiration, epididymal fluid passes through the glass micropipet, through the silastic tubing and into a plastic tuberculin syringe. Typically only microliters of fluid need to be retrieved. Sperm in the epididymal fluid are highly concentrated (roughly 1x106 sperm/ml) so this technique provides far more than an adequate number of sperm. Since post-thaw sperm viability is related to pre-freeze quality, it is worthwhile to retrieve and cryopreserve sperm of optimal quality to avoid the need for additional sperm retrieval procedures.
New Mini-micro-epididymal sperm aspiration (Nudell et al., 1998) is a combination of standard MESA and PESA procedures but uses a small incision that is well tolerated under local anesthesia. The procedure is performed with the spermatic cord block and sedation. A tunica vaginalis space entered via transverse 1-cm scrotal incision. A self-retaining eye-lid retractor is placed to create a ëwindowí into the tunica vaginalis space. The posteriorly located epididymis is rotated into view by gentle traction or placement of 7-0 Prolene traction suture into the epididymal tunic. Under the operating microscope the epididymal tunic is incised and individual tubule isolated. After careful hemostasis with bipolar cautery, epididymal tubule is incised tangentially with microscissors. Fluid is aspirated with a 24-gauge angiocath sheath attached to a 1.0-ml syringe. About 10 microliters of aspirated fluid is examined under 400X light microscope. Aspiration sites then progresses from cauda to caput in order to obtain the best quality sperm. Epididymal tubule and tunic are closed with 10-0 and 9-0 nylon sutures respectively. Tunica vaginalis space is closed with 4-0 absorbable suture after irrigation with saline and local anesthetic without epinephrine. Skin edges are reapproximated with 4-0 absorbable suture. The procedure was effective in 95% of 26 cases.
TEsticular Sperm Extraction (TESE)
Testicular sperm retrieval is usually timed to coincide with oocyte retrieval during a planned IVF cycle for the female partner. Under local or general anesthesia, sperm retrieval is effected using an open testicular biopsy technique. Scrotal exploration performed and testis is delivered into the wound in order to avoid any injury to the epididymis. Direct identification of testicular blood vessels is achieved using optical magnification with an operating microscope. Blood supply to the testis travels in a serpiginous fashion under the capsule (tunica vaginalis) of the testis. An avascular area close to midportion of medial, lateral or anterior surface of the testis is chosen, tunica albuginea incised with a 150 ultrasharp knife Retrieval of ample amounts of testicular parenchyma can be achieved with a single large incision in the tunica albuginea. The testicular parenchyma (approximately 500-mg) is excised with sharp, curved iris scissors and placed in HTF culture medium supplemented with 6% Plasmanate. Individual tubules are isolated by immediate initial dispersal of the specimen with 2 sterile glass slides. Additional mechanical dispersal performed by mincing of the tissue with sterile scissors in the HTF medium and performing sequential passes of tissue suspension through a 24-gauge angiocatheter. Wet preparation of this suspension is immediately examined under the phase contrast microscope at 100x and 400x power. Sampling is continued until spermatozoa are found or it is felt to be unsafe to perform additional biopsies. The incised segment of tunica albuginea is closed. Biopsy of the contralateral testis is performed if no sperm have been found. Multiple incisional biopsies should be avoided during TESE to prevent devascularization of the testis from damage to the subtunical testicular blood supply. It is also important to remember that sperm production is marginal in men with NOA; local inflammation and recovery after TESE may adversely affect sperm production for up to 6 months. Therefore, at least 6 months should elapse between TESE procedures. Dr. Peter Schlegel (Cornell University Medical Center) introduced new technique of microsurgical testicular sperm retrieval with isolation and retrieval of isolated seminiferous tubule.
Results of sperm retrieval (TESE) - ICSI for men with NOA have two different components. The first is the ability to retrieve sperm from the testis, and the second is the pregnancy rate after ICSI using those sperm. The chance of retrieving sperm has ranged up to 90% for men with NOA in a small series and as low as 50% in larger series. Obviously, the more careful selection of men before retrieval will determine the likelihood of finding sperm with TESE. Devroey et al. initially reported that 12 out of 15 (80%) men had sperm retrieved with TESE (80%). The fertilization rate per injected oocyte was 48%, with an overall clinical pregnancy rate of 20% (3/15) per attempt at TESE. Subsequent results from Belgium suggest that the rate of sperm retrieval is closer to 50% per TESE procedure. Schlegel et al. reported that 10/16 (62%) of men with NOA had sperm extracted with TESE. The fertilization rate per injected oocyte was 58%, and 5/10 (50%) of those undergoing ICSI achieved a clinical pregnancy [5/16 overall; (31% per TESE attempt.)] as standard therapy.
Cryopreservation of epididymal and testicular sperm
Successful sperm cryopreservation allows the opportunity for multiple ICSI cycles without the need for additional sperm retrieval procedures. Since ICSI enables even severely impaired sperm to effect oocyte fertilization, as long as sperm are viable, cryopreserved sperm can achieve acceptable rates of fertilization and pregnancy when coupled with ICSI. Cryopreservation also allows the potential for temporal separation of sperm retrieval procedures from assisted reproductive techniques. This is a significant benefit for the center that performs only occasional sperm retrieval procedures. If the cryopreserved sperm are viable after thawing, then this approach does not compromise the chance for achieving a pregnancy. Unfortunately, up to 10-20% of frozen epididymal samples will not have adequate viability after thawing for use with ICSI.